
Checking in on (RBQM) Risk Based Quality Management One Year After ICH E6(R3)
Risk Based Quality Management One Year After ICH E6(R3)
The industry is navigating a complex transition, balancing the adoption of innovative technology, maintaining study timelines, ensuring patient safety, and safeguarding data integrity. RBQM is a vital in helping with the adoption of these new technologies.
The risk-based approach is foundational to creating a data-driven culture for clinical trial management. By leveraging Artificial Intelligence (AI) driven capabilities for signal detection and trend analysis, teams can proactively identify potential issues, centralize oversight, and prevent delays, all while maintaining data quality.
On January 6, 2025, the International Council for Harmonization (ICH) adopted the long-awaited E6 (R3) Good Clinical Practice (GCP) guidelines. This reinforces the need to continue efforts to modernize trial conduct by emphasizing a flexible, risk-based approach to study management and data quality oversight.
The latest ICH E6 revision clearly specifies this shift:
Enhanced Risk Based Quality Management (RBQM): While E6 (R2) introduced Risk-Based Monitoring (RBM), E6 (R3) expands this to a comprehensive RBQM approach. It integrates proactive risk assessment at all trial stages (National Center for Biotechnology Information, 2024).
Bottom line: RBQM is a strategic imperative. By combining robust technology solutions with expert advisory support, organizations can transform clinical trial management into a proactive, adaptive, and compliant process aligned with ICH E6 (R3).
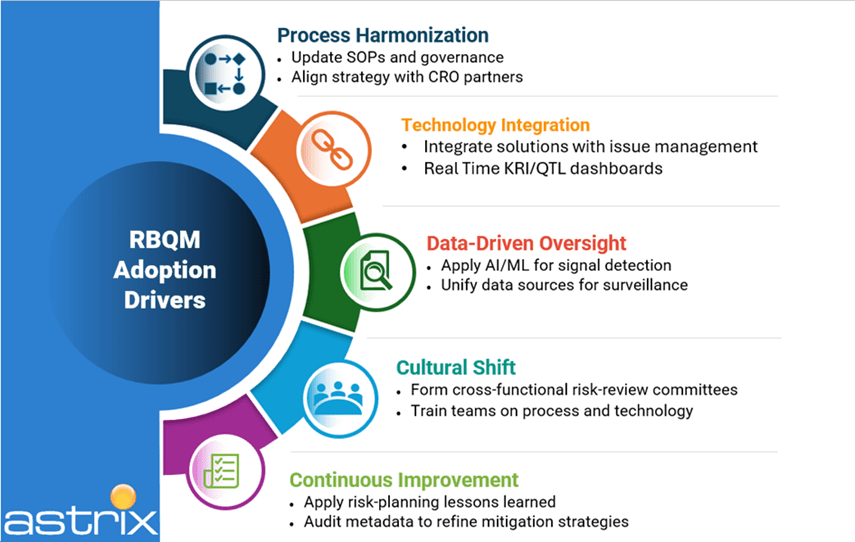
The first step toward holistic RBQM
The first step requires buy-in from key stakeholders involved in trial execution, including Biostatistics, Clinical Operations, Data Management, and Medical/Safety Monitoring. These roles provide critical expertise in risk identification and mitigation strategies.
Many organizations have adopted risk planning tools and collaborated with Contract Research Organizations (CRO) to address Key Risk Indicators (KRIs) and Quality Tolerance Limits (QTLs) throughout trial conduct. However, the full benefits of RBQM, such as centralized data quality oversight, reduced on-site monitoring, and accelerated timelines for data reviews and cleaning, are often unrealized. Why?
- Disparate Systems hinder the integration of data and issue management.
- Outdated Standard Operating Procedures (SOPs) do not align roles with this data-driven approach.
- Discordant Outsourcing Strategies limit the potential of RBQM.
What Obstacles Do Clinical Teams Face When Implementing a Risk-Based Approach?
The obstacles to implementing this approach stem from legacy processes, systems, and strategies. Automation and AI pinpoint anomalies and issues, empowering teams to act swiftly with targeted mitigations.
Legacy processes, trial management, and data capture systems require adaptation to effectively manage and surface risks, as well as capture mitigation and data quality oversight actions. Much of clinical trial management and oversight are outsourced to CROs. This is an efficient way to manage resource needs for trial monitoring and management; however, aligning it with the risk-based approach requires collaboration and mutual understanding of what this change entails.
Unlock success with Astrix’s proven blueprints. Consider consulting resources to guide your team in creating and executing an organizational change management plan. We use a proven methodology that enables clients and key team members to develop training and communication plans to meet adoption targets. This approach includes a phased rollout of pre-built training and processes and the needed resources to ensure your team members stay focused on their current workload.
Learn more about critical success factors for change management: Navigating Organizational Change
How Are Organizations Aligning Outsourcing Strategies with RBQM Solutions?
Many forward-thinking companies are moving to harmonize their outsourcing strategies with RBQM solutions by utilizing technology systems such as, CluePoints, Cyntegrity, Veeva RBSM, Medidata Clinical Data Studio, or TRI/Opra software systems.
These systems enable centralized risk planning, real-time monitoring, data quality oversight, and risk mitigation. They provide a collaborative environment for CRO resources to work with sponsors to effectively capture their oversight measures. While technology is a significant asset, success depends on updated governance frameworks, SOPs, and cultural alignment across teams.
Leverage trusted third-party advisory expertise to guide technology evaluation and adoption, align with RBQM methodology, and optimize collaboration with CRO partners. This support ensures Clinical Operations teams achieve seamless integration, effective governance, and adaptability across the spectrum of outsourcing models within clinical programs.
What are the benefits of RBQM Efficiencies for Study Timelines?
The primary benefit of RBQM is the centralization of clinical and operational data, enabling study teams to identify outliers, monitor oversight metrics, and maintain data integrity. This approach supports proactive data cleaning and review in parallel with data collection, reducing reliance on retrospective checks, accelerating decision-making, and ensuring submission-ready datasets with a clear record of ongoing risk-based reviews.
Expert Tip: Comprehensive data on how RBQM impacts key timelines such as the following remains limited:
- Final Protocol to Database Release
- Last Patient Last Visit to Database Lock
How can study teams evaluate RBQM’s impact on their study timelines?
Consider outsourcing to a consulting provider that has experience helping companies evaluate and implement RBQM solutions. These expert consulting services can help organizations define success criteria, benchmark critical milestones, and accurately assess the technology and organizational investments required to implement an effective RBQM strategy. The impact on study timelines is driven by your capacity to elevate data quality via timely corrective action.
What about Data Integrity?
Safety and efficacy data points now come from an increasing number of sources. This complex matrix of data, combined with dynamic trial designs that focus on event-driven assessments and bio-sampling, supports adaptive data quality oversight. This proven approach ensures data integrity and enables timely review of study-level risks during a clinical trial.
Consolidating data in a central platform aligned with predefined key risk indicators, quality tolerance limits, and critical-to-quality factors is the essential first step for effective RBQM. Astrix delivers frameworks for clinical data governance that empower teams with stakeholder-driven prioritization of data. These frameworks integrate timely oversight technology to mitigate risks as they arise and sustain data quality and integrity throughout the trial.
How can clinical teams incorporate risk-based approaches, considering the new guidance?
According to Gartner Hype Cycle, IT and Clinical Teams have voiced the following recommendations for adopting the risk-based approach/systems for clinical trials:
Evaluate RBM Tools: Organizations are encouraged to assess RBM tools that focus on identifying and mitigating risks, optimizing trial operations, and ensuring study, site, and data quality. Starting with tools provided by e-clinical platform vendors can help smooth the risk identification process.
Analytics-Based Tools: Consider analytics-based “bolt-on” RBM tools that can provide insights across multiple data sources, especially when collaborating with clinical research organizations or various e-clinical point solutions for trial operations (Hype Cycle for Life Science R&D, 2025)
Embracing RBQM for Future-Ready Clinical Trials
To fully realize these benefits, teams must move beyond basic risk assessments and integrate RBQM into their culture, processes, and technology stack. This requires:
- Cross-functional collaboration.
- Updated SOPs and governance frameworks.
- Integrated systems and advanced analytics.
The adoption of ICH E6 (R3) marks a turning point for clinical research, shifting from reactive monitoring to proactive, risk-based oversight. Organizations that embrace RBQM are not only ensuring compliance but also driving operational efficiency, reducing timelines, and improving data integrity.
About Astrix
Astrix is a market leader in delivering innovative solutions through world-class people, processes, and technology that fundamentally improve scientific outcomes and quality of life. Founded by scientists to address the unique challenges of life sciences and other science-based businesses, Astrix provides a growing array of strategic, technical, and staffing services that create value across organizations. Leveraging deep expertise in Clinical Operations and Regulatory Affairs, Astrix facilitates effective change by introducing innovative approaches to data quality oversight, ensuring compliance and operational excellence.
References
International Council for Harmonization. (2025). E6(R3) Good Clinical Practice: Final guideline. U.S. Food & Drug Administration. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/e6r3-good-clinical-practice-gcp
[fda.gov]
National Center for Biotechnology Information. (2024). R3 key improvements [PMC Article]. https://pmc.ncbi.nlm.nih.gov/articles/PMC12133055/
[nlm.nih.gov]
Harwood, R., & Smith, J. (2025, July 7). Hype Cycle for Life Science Enterprise Technologies, 2025. Gartner. https://www.gartner.com/en/documents/6687234
[gartner.com]
Case Study: LabWare Centralized Data Review for a Global Biopharmaceutical Company
Overview A global biopharmaceutical company specializing in discovery, development,... LEARN MOREWhite Paper: Managing Data Integrity in FDA-Regulated labs.
New White Paper LEARN MORELET´S GET STARTED
Contact us today and let’s begin working on a solution for your most complex strategy, technology and strategic talent services.
CONTACT US



