
5 Key Considerations Before Investing in eClinical Technology
What Private Equity Needs to Know About eClinical Investments
Private equity firms evaluating eClinical technology investments face a dynamic, innovation-heavy market. Automation, AI, and specialized LLM-driven solutions are emerging rapidly. While many new providers offer compelling capabilities, they may lack the scale, regulatory maturity, or proven customer adoption necessary for long-term success. Distinguishing true platform value from early-stage promise is essential for making confident investment decisions.
This article explores five critical factors private equity firms should consider before investing in an eClinical technology company. We will examine the market landscape, key value drivers, integration strategies, regulatory compliance, and the importance of a clear post-investment blueprint to help guide your due diligence process.
1. Understanding the eClinical Market Landscape
Industry analysis suggests that the global eClinical solutions market will grow rapidly, driven by increasing clinical trial complexity and ongoing efforts to improve data efficiency and operational performance. Business Research Insights (2026) expects the market to grow from USD 6.6 billion in 2026 to USD 12.97 billion by 2035, indicating a 7.8% CAGR, driven by increased trial complexity, demand for real‑time insights, and broader adoption of decentralized digital trial models.1
Systems for Electronic Data Capture (EDC), Clinical Trial Management (CTMS), Trial Master File (TMF), Clinical Outcomes Assessments (eCOA), and Regulatory Information Management (RIM) are central to this ongoing growth. For private equity firms, the core challenge is determining whether a target company’s technology can scale, integrate, and comply with evolving regulatory expectations. The market is fragmented, with established leaders and a consistent influx of niche innovators. A thorough analysis should assess a target’s competitive positioning, unique value proposition, and ability to adapt to industry trends, including decentralized clinical trials (DCTs) and the growing emphasis on real-world evidence (RWE).
2. Evaluating Core Technology and Architecture
A disciplined review of a platform’s technical, regulatory, and architectural maturity is fundamental to any eClinical technology evaluation. Superficial assessments can obscure significant underlying risks that may impact future value. An effective due diligence effort should center on the priority areas illustrated here, placing heightened focus on Data Architecture, Scalability and Performance, and Technical Debt:
Extensible Data Architecture
- An eClinical platform’s value primarily depends on its ability to manage and exchange data. A modern, extensible data architecture is crucial for supporting interoperability with other clinical, regulatory, and quality systems. Can the platform easily integrate with third-party applications via APIs? Does it adhere to industry data standards like CDISC? A flexible architecture enables greater connectivity across the clinical ecosystem, a significant value driver for end-users.
Scalability and Performance
- Assess whether the technology can manage growth. Does technology support an increasing number of users, larger data volumes, and more complex clinical trial protocols without performance degradation? Cloud-native solutions often provide greater scalability and flexibility than on-premises legacy systems. Scrutinize the platform’s infrastructure to understand its limitations and potential for expansion.
Technical Debt
- Many established platforms carry technical debt, which refers to the long-term costs associated with legacy code, outdated components, or brittle architectural decisions. High technical debt can hinder innovation, increase maintenance costs, and create security vulnerabilities. Identifying and quantifying this debt is critical for forecasting long-term supportability and calculating the full cost of ownership.
3. Assessing Regulatory Compliance and Quality Maturity
In the life sciences industry, regulatory compliance is non-negotiable. An eClinical platform must demonstrate robust alignment with global regulations to be a viable investment. This goes beyond simple documentation and requires a deep-seated culture of quality within the target organization.
Key Regulatory Frameworks
- The developers must design the technology to comply with critical regulations and guidelines, including Good Clinical, Laboratory, and Manufacturing Practices (GxP), the FDA’s 21 CFR Part 11, and the EU’s Annex 11. These regulations govern electronic records, electronic signatures, and computer system validation. During due diligence, confirm that the target company has a mature Quality Management System (QMS) and can provide evidence of validated systems and processes.
Data Integrity and Security
- Data integrity is paramount in clinical research. The system must have demonstrable controls to ensure data is attributable, legible, contemporaneous, original, and accurate (ALCOA+). Furthermore, with the rising threat of cyberattacks, robust security protocols are essential. Evaluate the platform’s security measures, data encryption standards, and disaster recovery plans to gauge its resilience.
4. Defining a Clear Integration and Value Creation Strategy
A successful investment hinges on a clear post-acquisition blueprint for value creation. The PE firm should develop this strategy during the due diligence phase and should identify specific opportunities to enhance the platform’s capabilities, market position, and financial performance.
The Post-Investment Blueprint
- A detailed blueprint should outline the steps for executing the investment thesis. These steps include identifying potential partnership or integration opportunities that could expand the platform’s functionality and market reach. For example, integrating an EDC system with an eCOA solution can create a more comprehensive offering for clinical trial sponsors. This plan should also include a technology roadmap that addresses any identified gaps and prioritizes future development efforts.
Executing the Strategy
- Executing the post-investment plan requires aligning the target company’s management with the strategic objectives. This may involve operational improvements, strategic tuck-in acquisitions, or expansion into new geographic markets. Having a clear 100-day plan and longer-term milestones helps ensure that the implementation of the value-creation strategy is effective and aligned with the investment goals. An experienced partner can help manage these end-of-the-end technology integration programs to accelerate returns.
5. The Importance of Objective Due Diligence
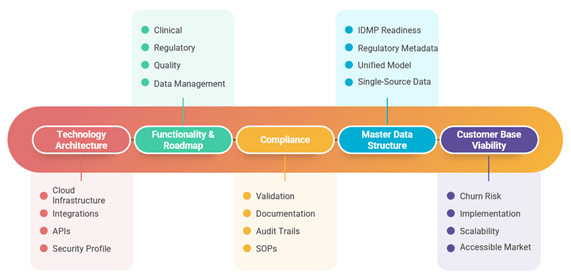
Given the complexity of the eClinical landscape, conducting rigorous, objective due diligence is crucial to mitigating risk and validating an investment thesis. Relying solely on a target company’s claims is insufficient. Employing a comprehensive assessment framework, as illustrated below, is needed to evaluate the technology, operations, and market position with an unbiased lens:
Using a structured framework with dozens of assessment categories can provide a holistic view of the target. This analysis should cover everything from the technology stack and architecture to the regulatory compliance posture and the strength of the management team. This process provides the fund manager with key outputs to assess the investment’s viability, including technology scalability forecasts, regulatory gap analyses, and clear value-creation pathways. This disciplined approach can turn clinical technology investments into high-performing portfolio assets.
What PE Firms Gain from Astrix’s Due Diligence Approach
Astrix’s evaluation provides fund managers with actionable insights that directly inform investment decisions and post‑close strategy:
- Partnership and integration opportunities
- Technology scalability forecasts
- Regulatory gap analysis and risk assessment
- Clear value‑creation pathways
Integration Strategy and Post‑Investment Execution
Using the Astrix Due Diligence Blueprint™, we identify risks and integration requirements across Clinical, Regulatory, Quality, and Data Repository systems. Astrix built our framework on more than 40 technology assessment categories and uses this to determine platform efficacy, scalability, and alignment with the investment thesis.
Astrix draws on decades of experience leading end‑to‑end vendor evaluations, system implementations, and enterprise transformations across the life sciences sector.
Why Private Equity Firms Partner with Astrix
Astrix brings a 30‑year history of delivering scientific, digital, and regulatory expertise across the life sciences value chain. Our team has deep experience leading eClinical and regulatory platforms, including platforms such as RIM, CTMS, EDC, TMF, eCOA, ICF, etc., from providers including Veeva, Medidata, Florence, and more.
- We have successfully led rigorous due diligence engagements and managed complex technology integration programs that accelerate value creation for PE‑backed portfolio companies. With a global delivery model spanning the U.S., Europe, and Latin America, we provide scalable, cost‑efficient execution tailored to fast‑moving investment environments.
Astrix turns eClinical technology investments into high‑performance, high‑return portfolio assets.
About Astrix
Astrix is the global leader in delivering innovative strategies, solutions, and talent to the life sciences industry. Powered by world-class people, proven processes, and advanced technology, Astrix partners with clients to drive measurable improvements in business performance, scientific advancement, and clinical outcomes—ultimately driving towards a goal of improving quality of life. Founded by scientists to address the industry’s most complex challenges, Astrix provides a growing portfolio of strategic and technical services that deliver immediate impact while enabling long-term digital transformation. Our deep expertise spans strategic planning, data strategy, AI/ML readiness and technologies, lab informatics, and modern clinical operations and eClinical platforms so we can successfully deliver solutions that have high impact and drive better outcomes for everyone.
Citations:
1Business Research Insights. (2026). EClinical solutions market 2026–2035: Growth at CAGR 7.8%. https://www.businessresearchinsights.com/market-reports/eclinical-solutions-market-126540
Case Study: LabWare Centralized Data Review for a Global Biopharmaceutical Company
Overview A global biopharmaceutical company specializing in discovery, development,... LEARN MOREWhite Paper: Managing Data Integrity in FDA-Regulated labs.
New White Paper LEARN MORELET´S GET STARTED
Contact us today and let’s begin working on a solution for your most complex strategy, technology and strategic talent services.
CONTACT US



